
Home
Common Compounds
Exam Guide
FAQ
Features
Glossary
Construction Kits
Companion Notes
Just Ask Antoine!
Simulations
Slide Index
Toolbox
Tutorial Index
Introduction
Measurement
| Matter |
Compounds
Chemical change
The mole
Gases
Energy & change
The quantum theory
Electrons in atoms
The periodic table
Chemical bonds
Solids
Liquids
Solutions
Acids & bases
Redox reactions
Reaction rates
Organic chemistry
Everyday chemistry
Inorganic chemistry
Environmental chemistry
Laboratory
History of chemistry
Miscellaneous
Home  FAQ FAQ  Matter Matter | Print | Comment |
| Previous Question | Next Question |
How is caffeine separated from coffee?
-
-
For most of the twentieth century, coffee was decaffeinated with dichloromethane, CH2Cl2. Dichloromethane selectively dissolves caffeine without carrying off sugars, peptides, and flavor ingredients, so it removes coffee's buzz without changing its taste. However, it is somewhat toxic, and when evidence suggested that it might be carcinogenic, its use was sharply curtailed.
Vocabulary caffeine 
critical point
critical pressure
critical temperature
dichloromethane
distillation
extraction
recrystallization
reverse osmosis
supercritical fluid
Ethyl acetate was used as a replacement for dichloromethane during the 80's and early 90's. Although it too is moderately toxic, coffee makers touted ethyl acetate as "natural" because it was present in fruit.
A nontoxic and more environmentally benign solvent than either dichloromethane or ethyl acetate is now used: supercritical fluid carbon dioxide.
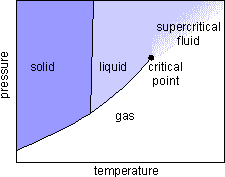 When a sealed vial containing both gaseous and liquid carbon dioxide under high pressure is heated, the liquid density drops while the gas density rises. If the pressure is above 72.8 atm, and the temperature rises above
304.2 K, the density of the liquid and the density of the gas become identical.
The meniscus between the liquid and gas phases vanishes. The carbon dioxide becomes a supercritical fluid
which has both gaslike and liquidlike properties. The fluid fills the container like a gas, but can dissolve substances like a liquid. Supercritical fluid carbon dioxide is an excellent nonpolar solvent for many organic compounds, including caffeine.
When a sealed vial containing both gaseous and liquid carbon dioxide under high pressure is heated, the liquid density drops while the gas density rises. If the pressure is above 72.8 atm, and the temperature rises above
304.2 K, the density of the liquid and the density of the gas become identical.
The meniscus between the liquid and gas phases vanishes. The carbon dioxide becomes a supercritical fluid
which has both gaslike and liquidlike properties. The fluid fills the container like a gas, but can dissolve substances like a liquid. Supercritical fluid carbon dioxide is an excellent nonpolar solvent for many organic compounds, including caffeine.
The extraction process is simple. Supercritical carbon dioxide is forced through green coffee beans. Its gaslike behavior allows it to penetrate deep into the beans, and it dissolves 97-99% of the caffeine present. The caffeine is valuable, and coffee manufacturers usually try to recover it. The caffeine-laden CO2 is sprayed with high pressure water and caffeine is then isolated by a variety of methods, including charcoal adsorption, distillation, recrystallization, or reverse osmosis.
References
- O'Brien, M.J., Spence, J.E., Skiff, R.H., Vogel, G. J., Prasad, R.: "Caffeine Recovery from Supercritical Carbon Dioxide", US Patent 4.996.317, 1991
Decaffeinating Coffee
(Scientific American) 
A brief article by Saul Katz describes various methods for decaffeinating coffee. The article includes a step-by-step illustration of supercritical fluid carbon dioxide extraction of caffeine.
http://www.sciam.com/0697issue/0697working.html (04/06/99)
Related resources
-
Introduction to Supercritical Fluids
(Phasex Corporation)
Supercritical Research Group
A brief introduction to supercritical CO2 extraction, an economically important technology used to decaffeinate coffee and tea, extract hops flavors in brewing, and extracting aromas and flavors from spices and herbs.
http://www.phasex4scf.com/scf.htm (7/2/99)(University of South Wales) 
An introduction to supercritical fluids and their applications, including decaffeination of coffee and waste treatment.
http://www.ceic.unsw.edu.au/centers/SF1/scf.htm (04/06/99) - O'Brien, M.J., Spence, J.E., Skiff, R.H., Vogel, G. J., Prasad, R.: "Caffeine Recovery from Supercritical Carbon Dioxide", US Patent 4.996.317, 1991
Decaffeinating Coffee
General Chemistry Online! How is caffeine separated from coffee?
Copyright © 1997-2010 by Fred Senese
Comments & questions to fsenese@frostburg.edu
Last Revised 02/23/18.URL: http://antoine.frostburg.edu/chem/senese/101/matter/faq/decaffeinating-coffee.shtml