
Home
Common Compounds
Exam Guide
FAQ
Features
Glossary
Construction Kits
Companion Notes
Just Ask Antoine!
Simulations
Slide Index
Toolbox
Tutorial Index
Introduction
Measurement
Matter
Atoms & ions
Compounds
Chemical change
The mole
Gases
Energy & change
The quantum theory
Electrons in atoms
The periodic table
Chemical bonds
Solids
Liquids
Solutions
Acids & bases
Redox reactions
Reaction rates
Organic chemistry
Everyday chemistry
Inorganic chemistry
| Environmental chemistry |
History of chemistry
Miscellaneous
Home  FAQ FAQ  Environmental chemistry Environmental chemistry | Print | Comment |
| Previous Question | Next Question |
What is lindane?
-
-
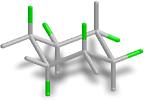 Lindane is 1,2,3,4,5,6-hexachlorocyclohexane, a musty-smelling crystalline solid used as an insecticide, especially to treat lice and scabies on the skin
of animals and humans. Of the many possible isomers, only the
Lindane is 1,2,3,4,5,6-hexachlorocyclohexane, a musty-smelling crystalline solid used as an insecticide, especially to treat lice and scabies on the skin
of animals and humans. Of the many possible isomers, only the  isomer shown at left has insecticide action. It is the active ingredient in many flea shampoos, soaps, and creams.
isomer shown at left has insecticide action. It is the active ingredient in many flea shampoos, soaps, and creams.
Lindane's use as an insecticide has declined because fleas and lice have developed resistance to it. Like most chlorocarbon pesticides, it degrades slowly in the environment and it accumulates in the fatty tissues of organisms at the top of the food chain. Many countries have banned or restricted its use. Lindane is no longer manufactured in the United States.
Lindane is toxic to humans as well as to pests. It is not water soluble but is soluble in fat and can pass directly through the skin. Toxic reactions range from mild skin irritation to dizziness, headaches, diarrhea, nausea, vomiting, and even convulsions and death. The most severe of these reactions is usually the result of accidental ingestion rather than skin contact. The estimated fatal dose is 150 mg/kg of body weight. Children and infants and pregnant women should avoid any contact with lindane.
References
-
The Merck Index
(Merck & Co.)
Lindane
This 2300 page book contains over 10000 compound entries. It is now available on CD ROM. The entries include chemical formulas and structures, physical properties such as density and solubility, references on syntheses, and toxicity and therapeutic category.
http://www.merck.com/faq/faq.html#mindex (11/12/98)(ExToxNet, Cornell University) 
A detailed profile of the pesticide lindane, including toxicological effects, ecological effects, environmental fate, exposure guidelines, and physical properties.
http://pmep.cce.cornell.edu/profiles/insect-mite/fenitrothion-methylpara/lindane/index.html (3/27/99)
General Chemistry Online! What is lindane?
Copyright © 1997-2010 by Fred Senese
Comments & questions to fsenese@frostburg.edu
Last Revised 02/23/18.URL: http://antoine.frostburg.edu/chem/senese/101/environmental/faq/what-is-lindane.shtml