
Home
Common Compounds
Exam Guide
FAQ
Features
Glossary
Construction Kits
Companion Notes
Just Ask Antoine!
Simulations
Slide Index
Toolbox
Tutorial Index
 |
 |
What do chocolate cravings, forgetful mice, and blissful pigs have in common? The answer is anandamide, a recently discovered messenger molecule that plays a role in pain, depression, appetite, memory, and fertility. Its name comes from ananda, the Sanskrit word for "bliss". Anandamide's discovery may lead to the development of an entirely new family of therapeutic drugs. Anandamide chemistry provides a rare glimpse of processes that affect human behavior at the molecular level.
The bliss receptor
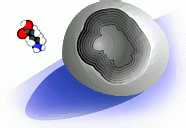
Nerve cells communicate by releasing special 'key' molecules that are intercepted by other nerve cells downstream. There are many different types of molecular keys, each with its own distinctive shape. The surfaces of the target nerve cells are studded with receptors, which are like locks to fit the keys. When the key fits into the receptor, the surface of the nerve cell changes in some way. For example, when the key molecule at right locks into the receptor on the surface of a nerve cell, it opens a door in the membrane that allows chloride ions to flood into the cell. This equalizes charges inside and outside the cell and prevents the cell from firing.
The keys must be removed again from the lock somehow, or the nerve cell will be permanently prevented from firing. Certain enzymes are produced that remove (by degrading and destroying) the keys after a certain amount of time, so that the nerve cell can go back to work.
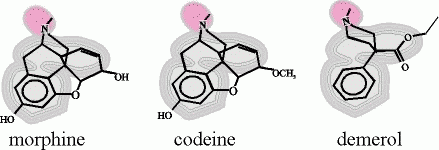
Drugs that have a powerful effect on the central nervous system often mimic natural molecular keys. For example, morphine is a potent pain killer that was found to lock into an 'opiate receptor' present on nerve cells.
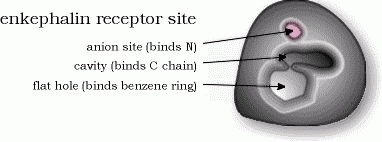
Scientists reasoned that since morphine is not naturally present in the body, there must be a natural key molecule with a very similar shape that activates this receptor. The natural keys turned out to be molecules called enkephalins. Although morphine is just a forgery of enkephalins, it's much more powerful (and more addictive) than the enkephalins because the key-removing enzymes can't pry it from the receptors. In time, less addictive forgeries (codiene and demerol) were introduced.
Scientists began to look for receptor sites to explain the action of other drugs and toxins in a similar way. In 1988, specific receptors were discovered for THC (tetrahydrocannibol, the active ingredient in marijuana).
THC isn't found naturally in the body. The existence of a specific 'bliss receptor' for THC implied that it was actually just a forgery of a hitherto unknown natural molecular key. The key was isolated by Israeli scientist Raphael Mechoulam in 1992: arachidonyl ethanolamide, later called 'anandamide':
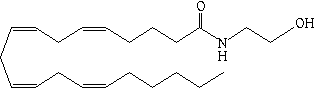
Anandamide's long hydrocarbon tail makes it fat-soluble and allows it to easily slip across the hydrocarbon-rich curtain that isolates the brain from the bloodstream. Notice that its three-dimensional shape strongly resembles that of THC. But unlike THC, anandamide is fragile. It breaks down very quickly in the body. That explains why anandamide doesn't produce a perpetual natural 'high'.
Forgetful mice and blissful pigs
Anandamide is synthesized enzymatically in areas of the brain that are important in memory and higher thought processes, and in areas that control movement. That implies that anandamide's function is not just to produce bliss.
Connections between nerve cells are associated with learning and memory. Nerve cells can make new connections and break old ones. Repeated use of a connection makes it grow stronger; lack of use can cause the connection to be lost. Some biochemical evidence suggests that anandamide plays a role in the making and breaking of short term neural connections [Derkinderen, 1996]. And animal studies suggest that anandamide induces forgetfulness. Substances that keep anandamide from binding to its receptor might be used to treat memory loss.
USDA agricultural researcher Gary Weesner is studying anandamide's effectiveness as an animal sedative [Pork, 1997]. "Pigs [treated with anandamide] walk less and lay down more," Weesner says. "We see reduced body temperature and slower respiration- all indicators consistent with being calm."
Chocolate and anandamide
Three compounds that strongly resemble anandamide were found in dark chocolate by Daniele Piomelli and co-workers at the Neurosciences Institute in San Diego [Piomelli, 1996]. They also found compounds (N-acylethanolamines) that block the breakdown of anandamide. Piomelli speculates that part of the pleasure of chocolate comes from anandamide and the anandamide-preserving N-acylethanolamines. "We are talking about something much, much, much, much milder than a high", he says.
The Nature article has been used by some to equate the effects of chocolate and cannabis to bolster arguments about the legalization of marijuana. "It is not that simple," Piomelli says. The response to THC and to the chocolate anandamides are not at all the same, even if the concentrations could be made comparable.
Piomelli was bemused by the spin his research was given in the popular press. "...You may be able to improve mood by blocking the breakdown of anandamide. It's not just something cute that we've done so that now we know more about chocolate. The hope is that it may contribute to helping cure mental disease," he said in a recent interview.[Psychiatric News, 1996]
Mom's first call?
Outside the brain, anandamide acts as a chemical messenger between the embryo and uterus during implantation of the embryo in the uterine wall. As such, it's one of the first communications that occurs between mother and child.
The highest concentrations of anandamide in the body were not in the brain, but in the uterus just before embryo implantation (at least, in the animal studies done so far) [KUMC, 1996] The concentration of anandamide changes as the uterus becomes more receptive to embryo implantation. The researchers were able to locate a definite target for the uterus' anandamide signal: mouse embryos contain more anandamide receptors than any tissue known, including the brain.
If THC can lock into anandamide receptors, there is the disturbing possibility that it may interfere with signaling between the uterus and the embryo. Indeed, 2-cell mouse embryos exposed to THC-like compounds have a significantly lower survival rate and exhibit a number of abnormalities [Yan, 1996]. More work will have to be done to see if these animal experiments mean that THC can interfere interfere with early pregnancy in humans.
A new key to the bliss receptor
Anandamide is not the only THC-like molecule used for signalling in the brain. Piomelli's group has recently reported a new molecular key that closely resembles anandamide [Piomelli, 1997]. Naturally produced sn-2 arachidonylglycerol (2-AG) can also lock into the bliss receptor. 2-AG is present at 170 times the concentration of anandamide in some regions of the brain. Piomelli speculates that 2-AG and anandamide perform complementary functions. Understanding how those functions work may allow some of the positive medicinal effects of anandamide and THC to be exploited therapeutically- without most of the negative effects.
Links and references
- Brain cannabinoids in chocolate
- E. di Tomaso, M. Beltramo, D. Piomelli, Nature, 382, 677-8 (1996).
- Coming: Drug therapy for chocoholics?
- Science News, 147, 374 (1996).
- Chocolate may mimic marijuana in brain.
- Chemical and Engineering News 74, 31 (1996).
- Researchers say chocolate triggers feel-good chemicals (CNN, Linda Ciampa)
- A CNN article that mentions Piomelli's chocolate anandamides. Feb. 14, 1996.
- Psychiatric News, November 1
- A recent interview with Daniele Piomelli.
- Home Page of William A. Devane (UW- Madison)
- Dr. William A. Devane, a molecular pharmacologist at the University of Wisconsin-Madison School of Pharmacy, is investigating natural enzymatic processes that produce anandamide. Devane hopes that detailed knowledge of the structure of anandamide-building enzymes may lead to insights into their role in several psychiatric diseases.
- Production: Natural chemical calms stressed-out pigs
- Anandamide is being used as an experimental veterinary sedative by U. S. Department of Agriculture researchers.
- Dale Deutsch (SUNY-Stony Brook)
- Dale Deutsch's research focuses on how anandamide levels in the brain are regulated. A biochemist at the State University of New York at Stony Brook, Deutsch has found several substances that block the breakdown of anandamide. These substances may eventually have therapeutic value in any area where anandamide plays a role.
- P. Derkinderen, M. Toutant, F. Burgaya, et. al., Science, v. 273 # 5282, Sept 20 1996 pp. 1719-1722
- Nature, 388, 773 (1997).
- Activation of brain-type cannabinoid receptors interferes with preimplantation mouse embryo development
- Z. M. Yan, B. C. Paria, S. K. Dey, Biol. Reprod., 55, 756-761 (1996).
- Anandamide Levels And Cannabinoid Receptors In The Mouse Embryo (KUMC)
- Studies of anandamide signalling in early pregnancy.
General Chemistry Online! The Bliss Molecule
Copyright © 1997-2010 by Fred Senese
Comments & questions to fsenese@frostburg.edu
Last Revised 02/23/18.URL: http://antoine.frostburg.edu/chem/senese/101/features/anandamide.shtml