
Home
Common Compounds
Exam Guide
FAQ
Features
Glossary
Construction Kits
Companion Notes
Just Ask Antoine!
Simulations
Slide Index
Toolbox
Tutorial Index
Introduction
Measurement
Matter
Atoms & ions
Compounds
| Chemical change |
Gases
Energy & change
The quantum theory
Electrons in atoms
The periodic table
Chemical bonds
Solids
Liquids
Solutions
Acids & bases
Redox reactions
Reaction rates
Organic chemistry
Everyday chemistry
Inorganic chemistry
Environmental chemistry
Laboratory
History of chemistry
Miscellaneous
Home  FAQ FAQ  Chemical change Chemical change | Print | Comment |
| Previous Question | Next Question |
What are some examples of reactions that involve catalysts?
-
-
Catalysts are substances that increase the rate of a reaction by providing a low energy "shortcut" from reactants to products. In some cases, reactions occur so slowly that without a catalyst, they are of little value. Nearly all reactions that occur in living cells require catalysts called enzymes- without them, life would be impossible.
Vocabulary alkane 
alkene
catalyst
equilibrium
heterogeneous mixture
homogeneous mixture
There are two important classes of catalysts: homogenous catalysts like enzymes and aqueous ions that are uniformly mixed with the reactants, and heterogeneous catalysts that provide a surface that holds and reconfigures the reactants in a way that is favorable for reaction.
Examples of homogeneous catalysis. Catalysts in solution with the reactants usually provide fast reaction paths by allowing reactants to form an unstable intermediate that quickly decomposes into products. For example, the substitution reaction
is catalyzed by acid because the ethanol is converted into unstable CH3CH2OH2+, which quickly reacts with Cl- to produce the products.CH3CH2OH(g) + HCl(g) H2SO4 
CH3CH2Cl + H2O(  )
)The iodide-catalyzed decomposition of hydrogen peroxide is another classic example of homogenous catalysis.
Enzymes are biological catalysts that combine with reactants (called substrates) to form an unstable intermediate. Enzymes are protein molecules folded into a complex shape that contains sites for capturing and tranforming substrates (active sites). Some enzymes contain embedded metal ions or other molecules that participate in the active site. An example is carbonic anhydrase, which contains a zinc ion. It catalyzes the reaction
H2CO3(aq)
The equilibrium lies far to the right, and rapid equilibration is essential to allow CO2 to diffuse out of the bloodstream and into the lungs. Enzymes and other catalysts never change the position of equilibrium; they only allow equilibrium to be established more rapidly. H2O(
H2O( ) + CO2(aq)
) + CO2(aq)
Unlike most inorganic catalysts, enzymes are reaction specific. They act only on particular substrates, which fit into their convoluted surfaces like a key fits a keyhole. (Some enzymes flex around the incoming substrate, and don't have a rigid "keyhole"). Also unlike most inorganic catalysts, enzymes are delicate, intricate molecular machines which can be damaged or destroyed by substances involved in the reaction they catalyze.
Examples of catalysis on surfaces. Metals can sometimes capture reactants on their surfaces and reconfigure them in a way that is favorable to reaction. Consider the reaction of alkenes with hydrogen to form alkanes. The reaction does not proceed at a measurable rate at room temperature, even though calculations predict that the equilibrium should lie far to the right. For example, ethylene can be hydrogenated using platinum as a catalyst.
The hydrogen molecule adsorbed onto the metal surface quickly dissociates into separate hydrogen atoms that skitter across the metal surface. When they encounter an adsorbed ethylene, they react with it quickly and easily because the energy required to break the H-H bond has already been expended. Some of the energy from the forming C-H bonds helps the product molecule leave (desorb) from the metal surface; some is released as heat and is absorbed by the metal. In essence, the metal provides a lower energy path for the reaction by efficiently coupling bond formation (which releases energy) with bond breaking (which requires energy).H2C=CH2(g) + H2(g) Pt 
H3CH-CH3(g) The ring-opening of cyclopropane can be catalyzed in a very similar way:
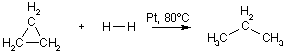
Platinum or vanadium(V) oxide catalyze the production of SO3 from SO2, a key step in the manufacture of sulfuric acid. Sulfuric acid is required for so many applications that annual production of the acid was used as an index of industrial productivity.
SO2(g) + 1/2 O2(g) V2O5 
SO3(g) Heterogeneous catalysis is used in automobile exhaust systems to decrease nitrogen oxide, carbon monoxide, and unburned hydrocarbon emissions. The exhaust is vented through a high-surface area chamber lined with platinum, palladium, and rhodium. For example, the carbon monoxide is catalytically converted to carbon dioxide by reaction with oxygen.
References and bibliography
- D. Dressler, H. Potter, Discovering Enzymes, W. H. Freeman, New York, 1991.
- A beautifully illustrated introduction to enzyme chemistry for readers with a general science background.
General Chemistry Online! What are some examples of reactions that involve catalysts?
Copyright © 1997-2010 by Fred Senese
Comments & questions to fsenese@frostburg.edu
Last Revised 02/23/18.URL: http://antoine.frostburg.edu/chem/senese/101/reactions/faq/examples-of-catalysts.shtml