
Home
Common Compounds
Exam Guide
FAQ
Features
Glossary
Construction Kits
Companion Notes
Just Ask Antoine!
Simulations
Slide Index
Toolbox
Tutorial Index
Introduction
Measurement
Matter
Atoms & ions
Compounds
Chemical change
The mole
Gases
Energy & change
The quantum theory
Electrons in atoms
The periodic table
Chemical bonds
Solids
Liquids
Solutions
| Acids & bases |
Reaction rates
Organic chemistry
Everyday chemistry
Inorganic chemistry
Environmental chemistry
Laboratory
History of chemistry
Miscellaneous
Home  FAQ FAQ  Acids and bases Acids and bases | Print | Comment |
| Previous Question | Next Question |
Is the salt of a weak acid and a weak base considered a weak acid or a weak base?
- Is the compound iodopyracet aka Diadrast(used to measure renal function) a weak acid or a weak base??? I cannot find this information anywhere!!
Bonnie 10/07/99 -
Here is the structure of iodopyracet, which is the salt of diethanolamine and 3,5-diiodo-4-pyridone-N-acetic acid:
Vocabulary amine 
amino acid
carboxylic acid
weak acid
weak base
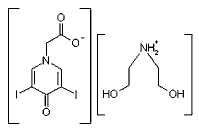
The carboxylate group can accept H+ ions. If the pH is low enough, the anion can act as a weak base. The NH2+ group can yield hydrogen ions, so if the pH is high enough, it can act as a weak acid. So the answer to your question is that (like amino acids) iodopyracet can behave either way.According to the Merck Index, aqueous iodopyracet solutions have a pH ranging from 5 to 8, depending on the concentration. Iodopyracet Injection (35% (w/v)), Diodrast Concentrated Solution (70% (w/v)), and Diodrast Compound Solution (40.5% (w/v), with 9.5% (w/v) of the diethylamine as well as the diethanolamine salt) are all neutral to litmus paper.
General Chemistry Online! Is the salt of a weak acid and a weak base considered a weak acid or a weak base?
Copyright © 1997-2010 by Fred Senese
Comments & questions to fsenese@frostburg.edu
Last Revised 02/23/18.URL: http://antoine.frostburg.edu/chem/senese/101/acidbase/faq/iodopyracet.shtml